|
mutLBSgeneDB |
| |
| |
| |
| |
| |
| |
|
| Gene summary for DDR2 |
 Gene summary Gene summary |
| Basic gene Info. | Gene symbol | DDR2 |
| Gene name | discoidin domain receptor tyrosine kinase 2 | |
| Synonyms | MIG20a|NTRKR3|TKT|TYRO10 | |
| Cytomap | UCSC genome browser: 1q23.3 | |
| Type of gene | protein-coding | |
| RefGenes | NM_001014796.1, NM_006182.2, | |
| Description | CD167 antigen-like family member Bcell migration-inducing protein 20discoidin domain receptor 2discoidin domain receptor family, member 2discoidin domain-containing receptor 2discoidin domain-containing receptor tyrosine kinase 2hydroxyaryl-protein | |
| Modification date | 20141207 | |
| dbXrefs | MIM : 191311 | |
| HGNC : HGNC | ||
| Ensembl : ENSG00000162733 | ||
| HPRD : 01868 | ||
| Vega : OTTHUMG00000034423 | ||
| Protein | UniProt: Q16832 go to UniProt's Cross Reference DB Table | |
| Expression | CleanEX: HS_DDR2 | |
| BioGPS: 4921 | ||
| Pathway | NCI Pathway Interaction Database: DDR2 | |
| KEGG: DDR2 | ||
| REACTOME: DDR2 | ||
| Pathway Commons: DDR2 | ||
| Context | iHOP: DDR2 | |
| ligand binding site mutation search in PubMed: DDR2 | ||
| UCL Cancer Institute: DDR2 | ||
| Assigned class in mutLBSgeneDB | B: This gene belongs to targetable_mutLBSgenes. | |
 Gene ontology having evidence of Inferred from Direct Assay (IDA) from Entrez Gene ontology having evidence of Inferred from Direct Assay (IDA) from Entrez |
| GO ID | GO Term | PubMed ID | GO:0018108 | peptidyl-tyrosine phosphorylation | 20004161 | GO:0038063 | collagen-activated tyrosine kinase receptor signaling pathway | 16186108 | GO:0046777 | protein autophosphorylation | 16186108 |
| Top |
| Ligand binding site mutations for DDR2 |
 Lollipop-style diagram of mutations at LBS in amino-acid sequence. Lollipop-style diagram of mutations at LBS in amino-acid sequence. We represented ligand binding site mutations only. (You can see big image via clicking.) |
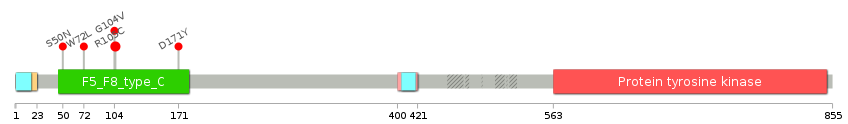 |
 Cancer type specific mutLBS sorted by frequency Cancer type specific mutLBS sorted by frequency |
| LBS | AAchange of nsSNV | Cancer type | # samples | R105 | R105C | COAD | 2 | W52 | S50N | COAD | 1 | R105 | R105C | KICH | 1 | C73 | W72L | LUAD | 1 | R105 | G104V | LUAD | 1 | H172 | D171Y | LUSC | 1 |
| cf) Cancer type abbreviation. BLCA: Bladder urothelial carcinoma, BRCA: Breast invasive carcinoma, CESC: Cervical squamous cell carcinoma and endocervical adenocarcinoma, COAD: Colon adenocarcinoma, GBM: Glioblastoma multiforme, LGG: Brain lower grade glioma, HNSC: Head and neck squamous cell carcinoma, KICH: Kidney chromophobe, KIRC: Kidney renal clear cell carcinoma, KIRP: Kidney renal papillary cell carcinoma, LAML: Acute myeloid leukemia, LUAD: Lung adenocarcinoma, LUSC: Lung squamous cell carcinoma, OV: Ovarian serous cystadenocarcinoma, PAAD: Pancreatic adenocarcinoma, PRAD: Prostate adenocarcinoma, SKCM: Skin cutaneous melanoma, STAD: Stomach adenocarcinoma, THCA: Thyroid carcinoma, UCEC: Uterine corpus endometrial carcinoma. |
 Clinical information for DDR2 from My Cancer Genome. Clinical information for DDR2 from My Cancer Genome. |
| DDR2 (discoidin death receptor 2) is a member of the DDR family of receptor tyrosine kinases that are stimulated by collagen rather than peptide growth factors. Downstream signaling in cancer cells is poorly understood but may be via SRC and STAT signaling pathways. Similar to integrin receptors, DDR2 may play a role in modulating cellular interactions with the extracellular matrix. DDR2 mutations have been found at low frequency in a number of cancers, including renal cell carcinoma, glioblastoma multiforme, endometrial cancer, colorectal cancer (COSMIC). The highest reported frequency is in squamous cell carcinoma of the lung (Hammerman et al. 2011). Related Pathways: Receptor tyrosine kinase/growth factor signaling. Paik, P.K. . 2015. DDR2. My Cancer Genomehttps://www.mycancergenome.org/content/gene/ddr2/ (Updated December 2015) |
| Top |
| Protein structure related information for DDR2 |
 Relative protein structure stability change (ΔΔE) using Mupro 1.1 Relative protein structure stability change (ΔΔE) using Mupro 1.1 Mupro score denotes assessment of the effect of mutations on thermodynamic stability. (ΔΔE<0: mutation decreases stability, ΔΔE>0: mutation increases stability) |
 : nsSNV at non-LBS : nsSNV at non-LBS : nsSNV at LBS : nsSNV at LBS |
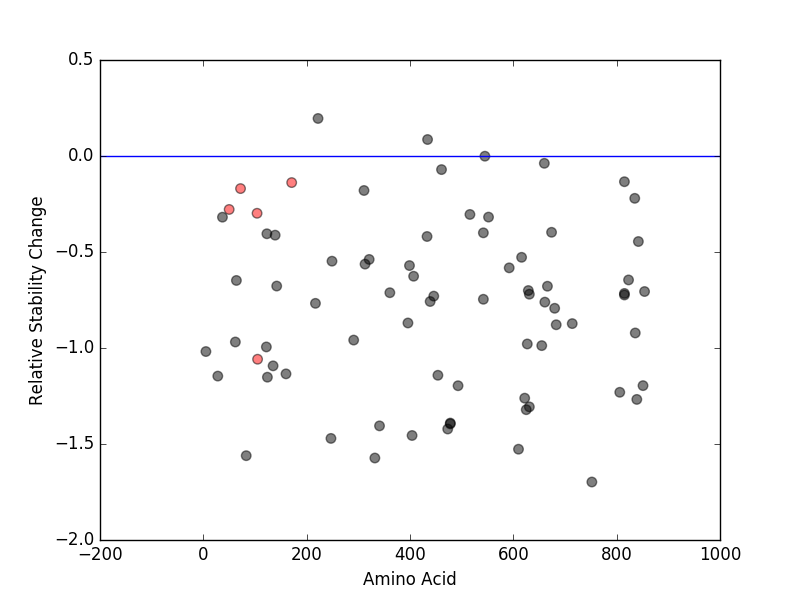 |
 nsSNVs sorted by the relative stability change of protein structure by each mutation nsSNVs sorted by the relative stability change of protein structure by each mutation Blue: mutations of positive stability change. and red : the most recurrent mutation for this gene. |
| LBS | AAchange of nsSNV | Relative stability change | R105 | R105C | -1.058482 | R105 | G104V | -0.29828989 | W52 | S50N | -0.27840686 | C73 | W72L | -0.16969475 | H172 | D171Y | -0.13842535 |
| (MuPro1.1: Jianlin Cheng et al., Prediction of Protein Stability Changes for Single-Site Mutations Using Support Vector Machines, PROTEINS: Structure, Function, and Bioinformatics. 2006, 62:1125-1132) |
 Structure image for DDR2 from PDB Structure image for DDR2 from PDB |
| PDB ID | PDB title | PDB structure | 2Z4F | Solution structure of the Discoidin Domain of DDR2 |  |
| Top |
| Differential gene expression and gene-gene network for DDR2 |
 Differential gene expression between mutated and non-mutated LBS samples in all 16 major cancer types Differential gene expression between mutated and non-mutated LBS samples in all 16 major cancer types |
 Differential co-expressed gene network based on protein-protein interaction data (CePIN) Differential co-expressed gene network based on protein-protein interaction data (CePIN) |
| Top |
| Top |
| Phenotype information for DDR2 |
 Gene level disease information (DisGeNet) Gene level disease information (DisGeNet) |
| Disease ID | Disease name | # PubMed | Association type |
| umls:C1849011 | Spondylometaepiphyseal Dysplasia, Short Limb-Hand Type | 1 | Biomarker, GeneticVariation |
 Mutation level pathogenic information (ClinVar annotation) Mutation level pathogenic information (ClinVar annotation) |
| Allele ID | AA change | Clinical significance | Origin | Phenotype IDs |
| Top |
| Pharmacological information for DDR2 |
 Gene expression profile of anticancer drug treated cell-lines (CCLE) Gene expression profile of anticancer drug treated cell-lines (CCLE)Heatmap showing the correlation between gene expression and drug response across all the cell-lines. We chose the top 20 among 138 drugs.We used Pearson's correlation coefficient. |
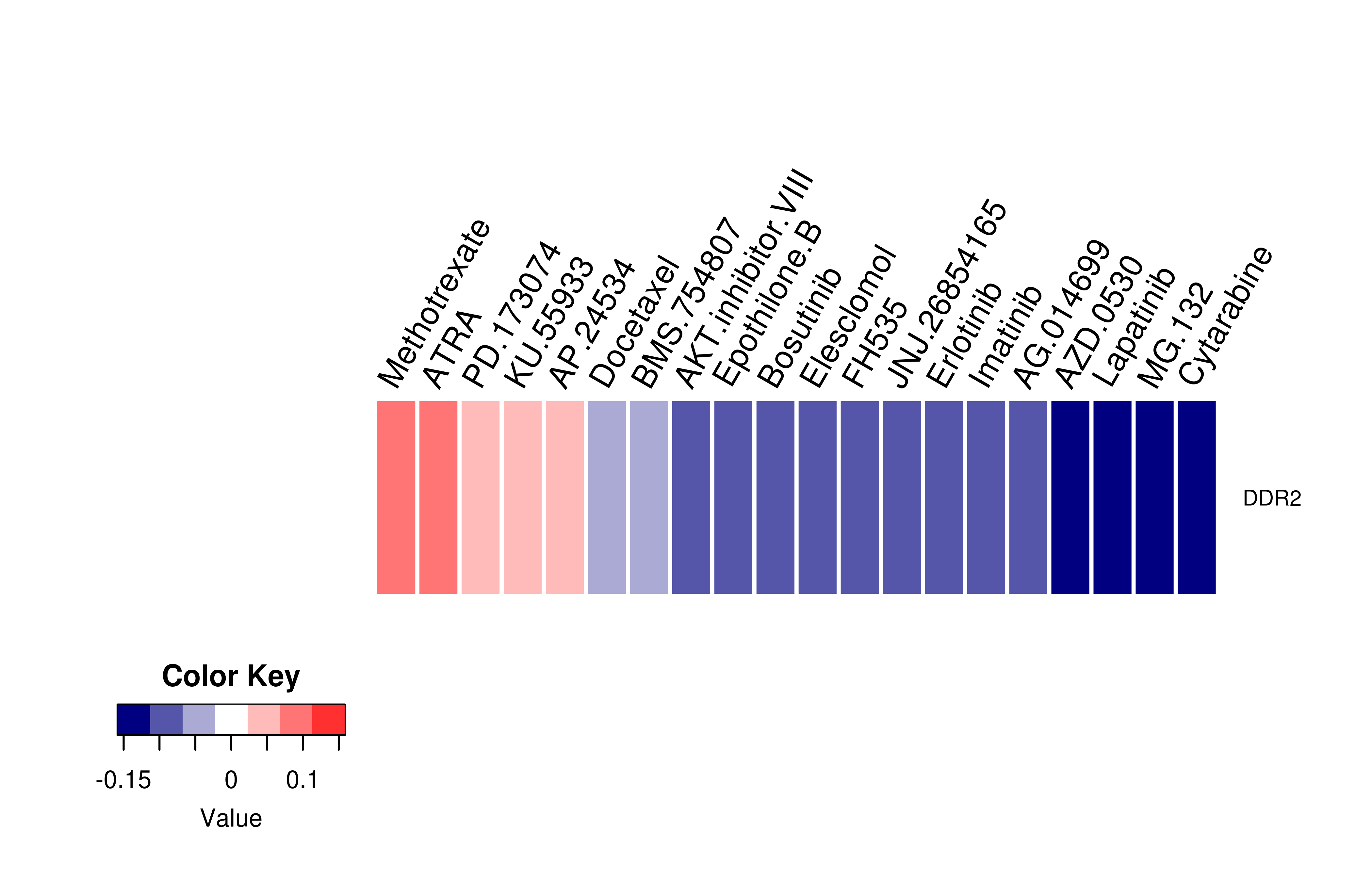 |
 Gene-centered drug-gene interaction network Gene-centered drug-gene interaction network |
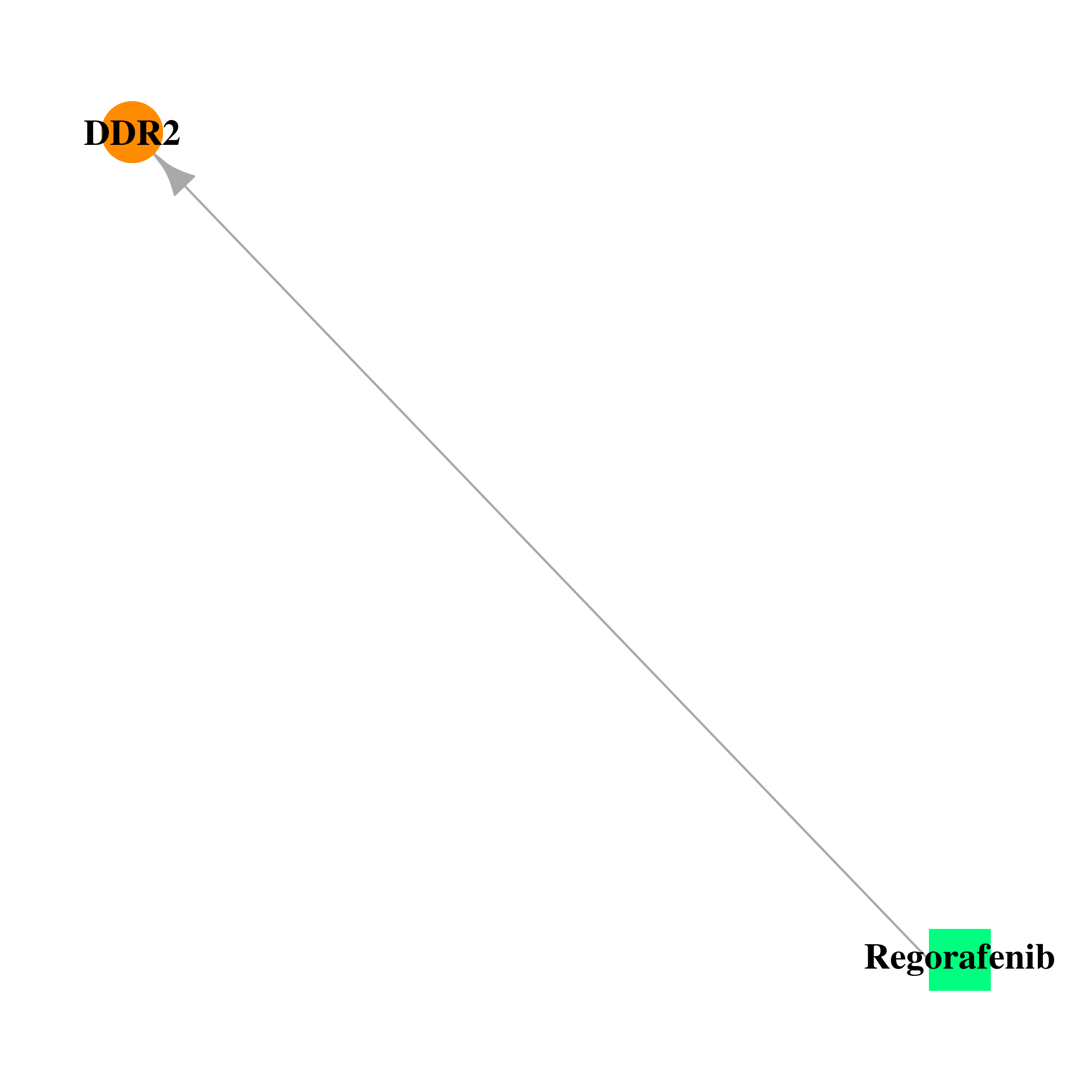 |
 Drug information targeting mutLBSgene (Approved drugs only) Drug information targeting mutLBSgene (Approved drugs only) |
| Drug status | DrugBank ID | Name | Type | Drug structure |
| Approved | DB08896 | Regorafenib | Small molecule | 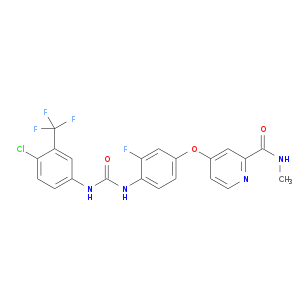 |
 Gene-centered ligand-gene interaction network Gene-centered ligand-gene interaction network |
 |
 Ligands binding to mutated ligand binding site of DDR2 go to BioLip Ligands binding to mutated ligand binding site of DDR2 go to BioLip |
| Ligand ID | Ligand short name | Ligand long name | PDB ID | PDB name | mutLBS | III | Peptide ligand (ACE,GLY,PRO,PRO,GLY,PRO,PRO,GLY,PRO,PRO,GLY,PRO,ARG,GLY,GLN,PRO,GLY,VAL,LEU,GLY,PHE,PRO,GLY,PRO,PRO,GLY,PRO,PRO,GLY) | 2wuh | A | W52 C73 R105 H172 | III | Peptide ligand (GLY,PRO,PRO,GLY,PRO,PRO,GLY,PRO,PRO,GLY,PRO,ARG,GLY,GLN,PRO,GLY,VAL,LEU,GLY,PHE,PRO,GLY,PRO,PRO,GLY,PRO,PRO,GLY) | 2wuh | A | W52 R105 |
| Top |
| Conservation information for LBS of DDR2 |
 Multiple alignments for Q16832 in multiple species Multiple alignments for Q16832 in multiple species |
| LBS | AA sequence | # species | Species | C177 | HSMNVCMRVEL | 2 | Homo sapiens, Mus musculus | C177 | STRTVCMRVEV | 1 | Caenorhabditis elegans | C73 | GDGAWCPEIPV | 2 | Homo sapiens, Mus musculus | C73 | GSGAWCPKNQI | 1 | Caenorhabditis elegans | D69 | DSEEGDGAWCP | 2 | Homo sapiens, Mus musculus | D69 | HQESGSGAWCP | 1 | Caenorhabditis elegans | E113 | GGHGIEFAPMY | 2 | Homo sapiens, Mus musculus | E113 | DGRGMEYATAF | 1 | Caenorhabditis elegans | H110 | RHAGGHGIEFA | 2 | Homo sapiens, Mus musculus | H110 | RFDDGRGMEYA | 1 | Caenorhabditis elegans | H172 | IPVTDHSMNVC | 2 | Homo sapiens, Mus musculus | H172 | VPVSNSTRTVC | 1 | Caenorhabditis elegans | I112 | AGGHGIEFAPM | 2 | Homo sapiens, Mus musculus | I112 | DDGRGMEYATA | 1 | Caenorhabditis elegans | N175 | TDHSMNVCMRV | 2 | Homo sapiens, Mus musculus | N175 | SNSTRTVCMRV | 1 | Caenorhabditis elegans | R105 | VGTQGRHAGGH | 2 | Homo sapiens, Mus musculus | R105 | VETQGRFDDGR | 1 | Caenorhabditis elegans | S173 | PVTDHSMNVCM | 2 | Homo sapiens, Mus musculus | S173 | PVSNSTRTVCM | 1 | Caenorhabditis elegans | T56 | WS-ESTAAKYG | 2 | Homo sapiens, Mus musculus | T56 | FDLQSTGPQHA | 1 | Caenorhabditis elegans | W52 | TASSQWS-EST | 2 | Homo sapiens, Mus musculus | W52 | SASSSFDLQST | 1 | Caenorhabditis elegans |
 |
Copyright © 2016-Present - The University of Texas Health Science Center at Houston |
